Professor Liu Long’s team from the Key Laboratory of Glycochemistry and Biotechnology, Ministry of Education, Jiangnan University published an article in “Applied microbiology and biotechnology”: “Combinatorial metabolic engineering enables high yield production of α‑arbutin from sucrose by biocatalysis”, through knockout and sucrose hydrolysis The related enzyme gene increases the conversion rate of sucrose to 51.5%. An inducible protein degradation system was also developed using Lon protease and a proteolytic tag to control the activity of PfkA, thereby allowing more fructose-6-phosphate to be converted to glucose-1-phosphate for α-arbutin synthesis.
And by integrating another copy of 6-phosphate glucose isomerase and phosphoglucolytic enzyme, the pathway from F6P to Glc1P was strengthened, a high α-arbutin titer was obtained, and the sucrose conversion rate increased to 60.4%.
In this study, substrate utilization was enhanced due to attenuated hydrolysis and the assistance of intracellular enzymes that reconvert by-products into substrates for α-arbutin synthesis. This strategy provides a new avenue for the whole-cell biocatalytic synthesis of other products using sucrose as a substrate, especially valuable sugars.
α-Arbutin has previously been produced by chemical synthesis and biotransformation. Among them, the chemical synthesis of α-arbutin requires protection and deprotection of OH groups due to harsh reaction conditions and complicated reaction process. In contrast, the biotransformation of α-arbutin was considered as a potential candidate. Because of its simpler synthetic procedures, mild reaction conditions, and less pollution, including enzymatic conversion, microbial fermentation, and whole-cell biocatalysis.
Whole-cell biocatalysis of α-arbutin was performed by engineered microbial cells expressing glycosyltransferases from different sources. They all use hydroquinone as an acceptor substrate, whose phenolic hydroxyl group can form α-isoglycosidic linkages with sugar groups from different donor substrates. Hydroquinone is easily oxidized under light to form dark benzoquinone without the ability to accept glycosides, and then generate oxygen free radicals to inhibit the catalytic activity of enzymes.
Whole-cell biocatalysis has several advantages: simple cell post-processing, high synthetic yield, and simple product isolation and purification. In addition, whole-cell catalysts can be recovered by simple centrifugation, thus reducing the cost of industrial production. However, in the previous whole-cell biocatalytic production of arbutin, the conversion rate of sucrose to α-arbutin was very low.
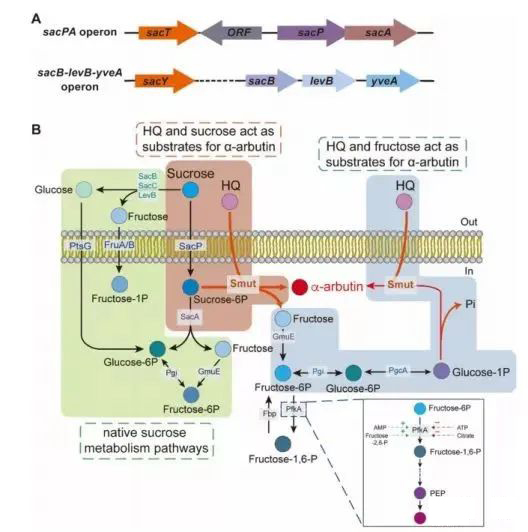
Figure 1. Operations involved in sucrose metabolism and the α-arbutin synthesis program in Bacillus subtilis.
Blocking the extracellular hydrolysis pathway of sucrose
During the growth of Bacillus subtilis, the two-region sacB-levB transcript and its products levansucrase and endolevanase all participate in extracellular sucrose metabolism in a certain order. Extracellular sucrose can induce the production of secreted levansucrase and membrane-anchored endolevanase, which then degrade sucrose into glucose and fructose extracellularly. Neither is a suitable donor substrate in the biotransformation of α-arbutin. Therefore, this added sucrose is wasted.
To avoid the extracellular breakdown of sucrose, the sacB, sacC, and levB genes were knocked out to generate recombinant strains BS8S401, BS8S402, and BS8S403, respectively (Fig. 2A). Knockdown of sacB, sacC, and levB genes had no appreciable effect on cell growth (Fig. 2B), whereas BS8S4 depleted sucrose within 24 h (Fig. 2C). The results showed that knocking out genes related to extracellular sucrose metabolism could significantly reduce sucrose consumption during cell growth.
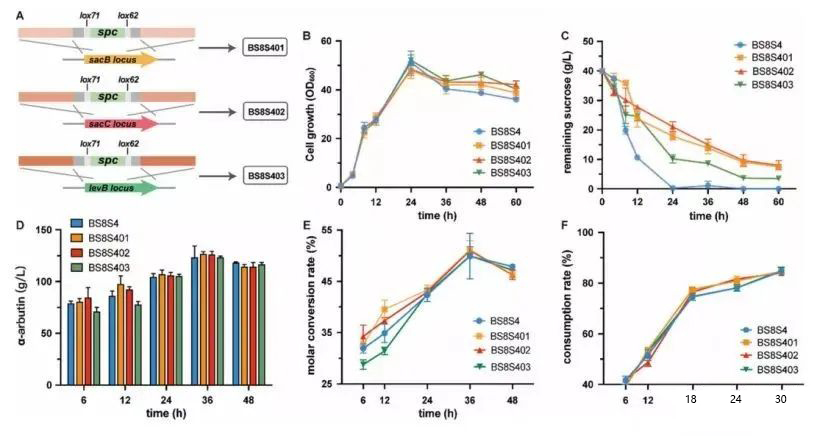
Figure 2. Effect of deletion of important genes in the extracellular sucrose breakdown pathway on α-arbutin production and sucrose consumption in recombinant Bacillus subtilis strains. A: Schematic diagram of knockout of sacB, sacC and levB genes. BS8S401 was obtained by deleting the sacB gene in BS8S4; this strain was used as the starting strain for expressing Smut in Bacillus subtilis WB800. Likewise, BS8S402 was obtained by deleting the sacC gene in BS8S401, and BS8S403 was obtained by deleting the levB gene in BS8S402. B: Cell growth curve of the recombinant strain. The vertical axis is the remaining sucrose concentration in the medium. C: Comparison of the sucrose utilization ability of different recombinant strains. D-F: Comparison of α-arbutin titer and sucrose consumption of different recombinant strains.
Blocking the intracellular competition pathway for sucrose
Sucrose is transported into cells via the PTS pathway and converted to sucrose-6-phosphate. The sacA-encoded sucrose-6-phosphate hydrolase competes with Smut for the same substrate (sucrose-6-phosphate).
To increase the conversion rate of sucrose-6-phosphate to α-arbutin, the sacA gene of BS8S401 was knocked out, resulting in the recombinant strain BS8S404 (Fig. 3A).
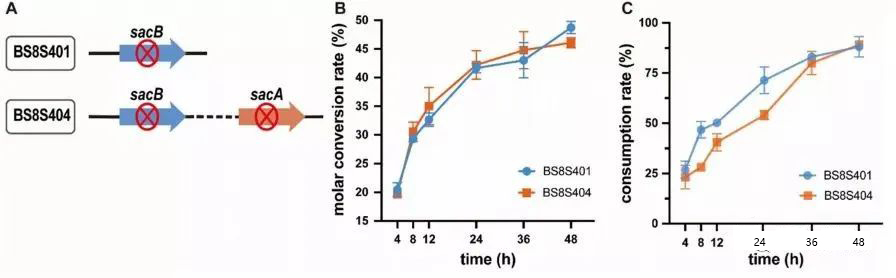
Figure 3. The effect of sacA gene deletion on the biotransformation of α-arbutin. A: BS8S404 was obtained by deleting the sacA gene of BS8S401. B: Comparison of sucrose conversion rates of different recombinant Bacillus subtilis. C: Comparison of consumption rates of different recombinant Bacillus subtilis.
BS8S401 and BS8S404 were cultured for whole-cell biocatalytic preparation, and the experimental results showed that the deletion of the sacA gene did not interfere with the growth of the cells. During the whole-cell bioassay, the sucrose consumption of BS8S404 cells was reduced by 17.4% compared with BS8S401 within 24 hours (Figure 3B), indicating that the inactivation of SacA prevents the hydroxylation of sucrose-6-phosphate, forming additional sucrose in α-Arbutin was consumed in the bioanalysis. However, within 24-48 hours, the sucrose consumption rate of BS8S404 cells gradually increased and approached that of BS8S401 cells, and the sucrose conversion rate finally reached 51.5%.
Regulation of pfkA expression by a protein degradation tag
As a by-product of engineering Bacillus subtilis to synthesize α-arbutin, fructose can be converted into G1P, which is another glycosyl donor substrate of Smut, in order to improve the carbon atom conversion efficiency of α-arbutin sucrose, aiming to reduce phosphate fructokinase activity to save more F6P for α-arbutin synthesis (Fig. 1B).
Bacillus subtilis can efficiently utilize glycerol as an ideal carbon source to generate intermediate glyceraldehyde-3-phosphate, which subsequently bypasses PfkA to enter the downstream glycolysis and TCA cycle (Fig. 4A). Therefore, the pfkA gene was knocked out, generating recombinant strain BS8S405. The growth of BS8S405 was significantly inhibited.

Figure 4. Glycerol utilization pathway of Bacillus subtilis and engineering strategies for tuneable PfkA degradation.
In addition, the researchers used a protein degradation tagging strategy to regulate PfkA expression and degradation (Fig. 4B) to ensure sufficient cellular biomass. The system consists of a polypeptide tag fused to the C-terminus of PfkA and xylose-induced MfLon. Thus, constitutively expressed PfkA can be degraded in a PDT-dependent manner.
The recombinant strain BS8S406 was constructed by inserting the mfon gene into the genome of BS8S404, and the strain could express MfLon by adding xylose. The candidate PDTs were fused to the C-terminus of PfkA using the CRISPR/Cpf1 system. Four plasmids were constructed, each constituting BS8S406. Finally, recombinant strains BS8S406-PWT and BS8S406-P3 were obtained (Fig. 4B).
Summarize
In this study, the researchers developed BS8S407 for whole-cell biocatalysis of α-arbutin with high sucrose conversion.
The findings suggest that combinatorial engineering of exogenous catalytic enzymes and host cell endogenous metabolic pathways can serve as an efficient and prospective strategy for whole-cell biocatalysis to increase substrate conversion rates. However, there are still some deficiencies, such as the proposed sucrose consumption in the later stages of biocatalysis.
In the future, scientists could further try to delete YugT, as it is hypothesized to act as an α-glucosidase, to prevent sucrose-6-phosphate hydrolysis during whole-cell biocatalysis. In addition, the expression levels of pgi and pgcA can be further increased by inserting more copies, and the adaptability of Smut to Glc1P can be improved through enzyme modification strategies, so that the conversion of F6P to Glc1P is more sufficient. Finally, the promoter for xylose-induced mfon gene expression can be replaced by a phase-specific promoter or an auto-inducible promoter to facilitate industrial applications.
