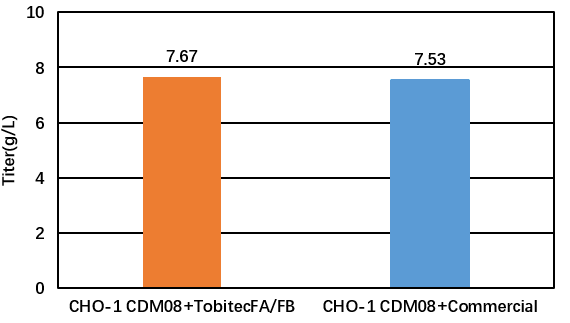CHO cells are widely used in the field of biopharmaceuticals
Chinese hamster ovary (CHO) cells are the first biopharmaceutical production cells approved by the US FDA, the EU EMA and the Chinese CFDA. They have been widely used in the large-scale preparation of various therapeutic protein drugs, including Recombinant antibodies, recombinant monomeric proteins and recombinant fusion proteins, etc. At present, scientists have successfully isolated different subtypes of CHO cell lines, such as CHO-K1, CHO-S, CHO DXB11, CHO DG44, CHO-M, and GS gene knockout CHO cells that have received continuous attention in recent years.
CHO-K1 was one of the first cell lines cloned from primary hamster ovary tissue and is still being studied by the biopharmaceutical industry and biopharmaceutical institutions. CHO-K1 are unmodified wild-type CHO cells. The most primitive CHO-K1 cells are adherent culture, and need to add serum, due to the stability of serum between batches, and later virus safety issues, serum-free suspension culture has become a trend. The expression platform based on CHO-K1 cells mostly adopts GS (glutamine synthetase) screening system and/or antibiotic screening system.
CHO protein production common culture model culture method
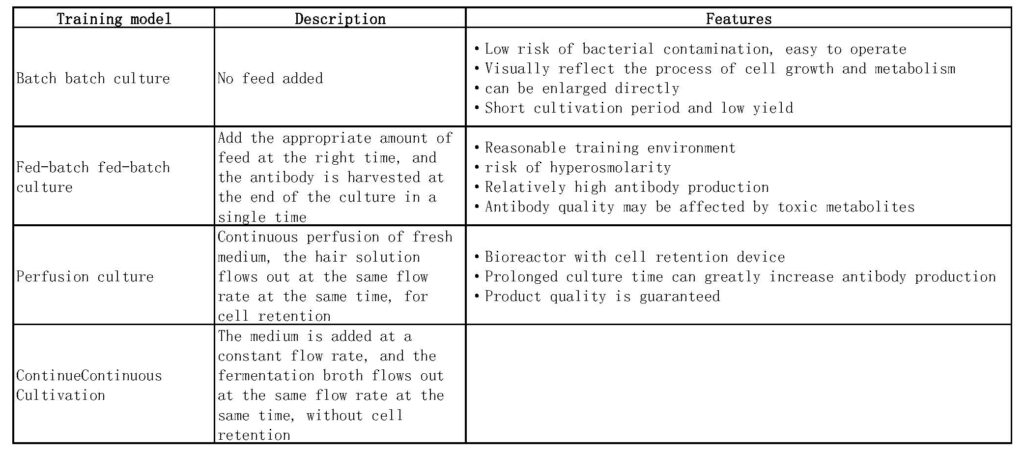
CHO-K1 cell culture operating procedures
01 Operating instructions
Ⅰ. Standard parameters
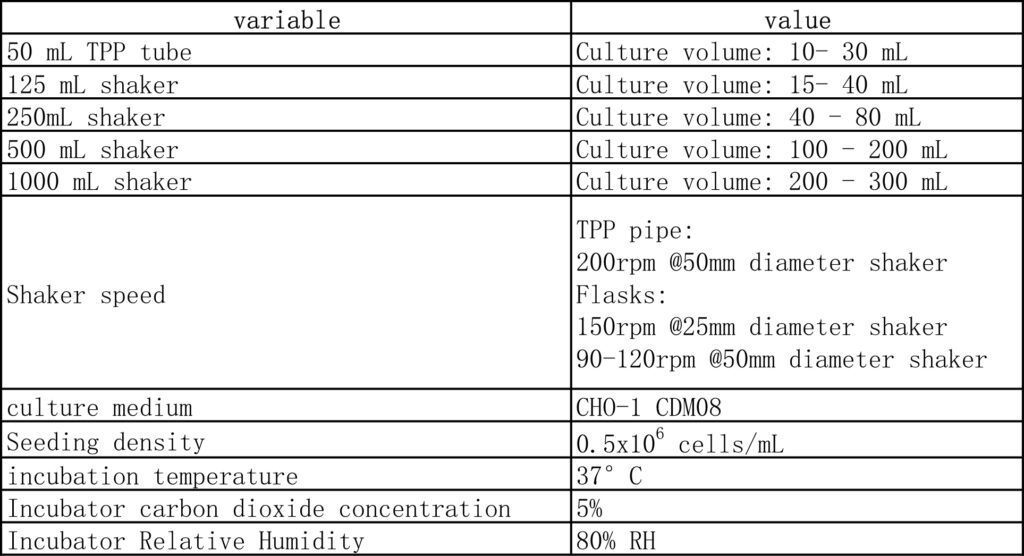
Ⅱ. Adaptation of culture medium
Under normal circumstances, there is no need to adapt the medium, and it can be directly replaced with CHO-1 CDM08. If direct replacement cells do not grow well, CHO-K1 cells grown in the original medium need to be transferred to a new medium consisting of 50% original medium and 50% culture (CHO-1 CDM08) for adaptive growth.
Ⅲ. Cell Subculture and Expansion
1. Preheat the CHO-1 CDM08 medium in a 37°C water bath.
2. Use 75% alcohol to clean/disinfect the biological safety cabinet.
3. Spray the medium bottle with 75% alcohol and place it in a biological safety cabinet.
4. Take out the shaker flask (or TPP tube) from the incubator, spray 75% alcohol and place it in a biological safety cabinet.
5. Take 0.5mL of cell suspension, and use a cell counter to analyze the viable cell density (×106 cells/mL) and viable cells (%).
6. Before passage, if the cell density is lower than 2.0 × 106 /mL, or the cell viability is lower than 80%, centrifuge at 150g ~ 300g (about 800rpm ~ 1200rpm) for 5 minutes to process the cells. Gently discard the supernatant, use 100% fresh medium to resuspend the cells at 0.5×106 /mL, inoculate the cells into a new shake flask (or TPP tube), and culture the cells according to the specified environmental conditions after passage (see section 1 standard parameter).
7. If the cell density before subculture is greater than 2.0×106/mL and the viable cell rate is higher than 85%, transfer an appropriate amount of cell suspension to a new shake flask (or TPP tube), and directly adjust the final culture volume with fresh medium Subculture the cells, and then culture the cells under the specified environmental conditions (see section 1 for standard parameters).
*This means that if the passaging ratio (seed density after passaging: cell density before passaging) is greater than 1:4, cells need to be centrifuged and resuspended in 100% fresh medium.
Ⅳ. Cell cryopreservation
1. Before freezing the cells, place the required materials such as the programmed cooling box containing fresh isopropanol, cryopreservation tubes, etc. in a 4°C refrigerator, pre-cool for at least 24 hours and then freeze them, or store them at -20°C for 2 to 4 hours. Store frozen after hours.
2. Sampling and counting, detection and analysis of viable cell density (×106 cells/mL) and cell viability (%).
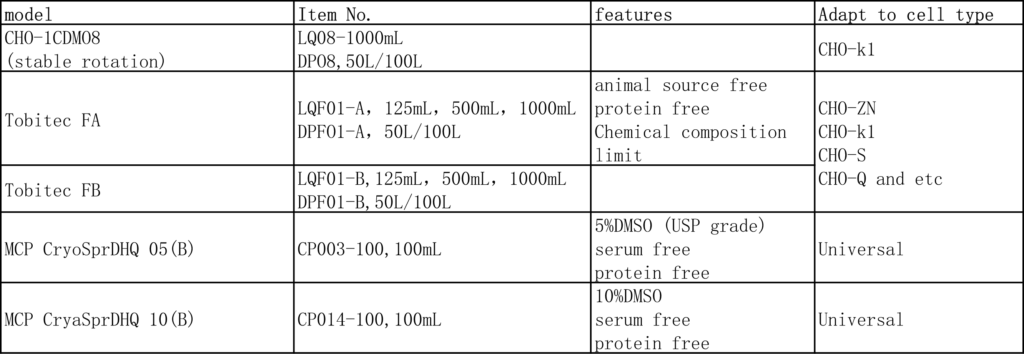
3. Use the cell freezing solution containing 10% DMSO (Tofflon product model: MCP CryoSpr-DHQ10 (B)) for freezing.
4. According to the number of frozen cells, frozen storage volume and frozen storage density (recommended freezing storage density is 10 ~ 20×106 /mL/vial), calculate the required amount of cells, and transfer to a centrifuge tube, at 800rpm ~ 1500rpm (ie 150g~300g) centrifuge for 5min.
5. Discard the supernatant, pat the bottom of the centrifuge tube to disperse the cells evenly, add growth medium with half the frozen storage volume, and gently pipette the cells to form a uniform cell suspension, see the table below (example and which one to choose according to the passage data) culture medium).
6. Use a sterile pipette to transfer the same volume of cell cryopreservation solution, and add it dropwise to the cell suspension. When adding the cell freezing solution, shake the centrifuge tube gently to keep the cell density uniform.
7. Use a disposable sterile pipette to transfer 1mL ~ 1.5mL cell suspension into a pre-frozen storage tube and store it on ice;
8. The frozen cell suspension needs to be mixed repeatedly during the aliquoting process. After aliquoting, transfer the cryovials to a pre-cooled programmed cooling box and store in a -80°C refrigerator for at least 24 hours.
9. Transfer to a liquid nitrogen tank for further storage.
Ⅴ. Precautions
·Cells are passaged in time ✔
·Media exposed to long-term light or heat ✘
·Cultivate at 2~8℃, keep away from light ✔
02 Supporting products
In order to improve the growth and production performance of CHO-K1 cells and meet the nutritional needs of high-density cell culture and high product expression, Tobitec FA & Tobitec FB feed medium can be added while using CHO-1 CDM08 medium. Combined with the characteristics of cell lines, a reasonable feeding strategy can significantly improve the growth of cells and the expression and quality of antibodies. The chemical composition of the feed medium is limited, does not contain any animal-derived components, and does not contain growth factors, polypeptides, hydrolyzates and phenol red. It is a high-efficiency feeding formula specially designed to improve the growth and production performance of CHO cells. It is suitable for the cultivation of different cell lines such as CHO-K1, CHO-S, DG44, and CHO-ZN using GS system and DHFR screening system.
03 Case of application of CHO-1 CDM08 in monoclonal antibody production
CCM: CHO -1 CDM08 +Tobitec FA&FB /Commercial
Cell line: CHO-K1
Process: feeding
Product Type: PD-1
PQAs: CQAs (key quality attributes), Titer (expression)
Figure 1: TOBITEC FA/FB feed medium protein expression test (PD-1)
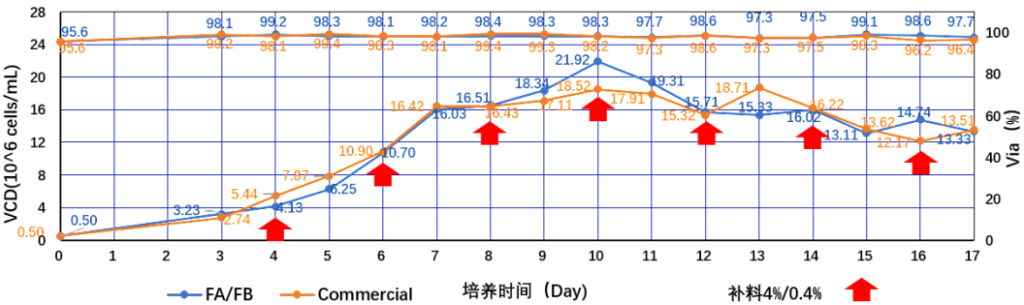
Figure 2: TOBITEC FA/FB feed medium protein expression test (PD-1)