The fragrance of jasmine is fresh, elegant, long-lasting and charming. Its unique fragrance is deeply loved by people and is known as one of the most popular floral fragrances in the world. However, traditional extraction methods are limited by high cost, low yield and seasonal factors, which greatly limits the wide application of jasmine fragrance in perfume, cosmetics and other industries, thus restricting the development of related industries.
Benzyl acetate, as the core component of jasmine fragrance, has a unique and rich aroma and is a key raw material in many industries. It is not only widely used in daily chemicals, paints, textiles and other industries, but is also an important component of soaps and flavors, and can even be found in some food flavors. However, the limitations of traditional extraction methods make it difficult for the production of benzyl acetate to meet market demand, limiting its application in various fields.
Currently, benzyl acetate is mainly produced through petroleum-based chemical synthesis methods, such as acetoxylation of toluene or benzylation of acetate using inorganic catalysts. Although this method has high production efficiency and low cost, its potential safety hazards and potential impact on the environment cannot be ignored.
In order to break through this bottleneck, the flavors and fragrances industry began to actively explore new production methods. With the rapid development of biotechnology, the use of engineered microorganisms to produce flavor substances has become a feasible alternative. Engineered microorganisms can simulate natural biosynthetic processes and produce purer, closer to natural products. Through an enzymatic transesterification reaction, engineered microorganisms can utilize benzyl alcohol and acyl donors such as vinyl acetate and ethyl acetate to efficiently produce benzyl acetate. This production method not only significantly improves production efficiency, but also reduces the impact on the environment.
Recently, two scientists from Groningen, the Netherlands, reported a new method adopted by Choi et al., which is to adopt a “delayed co-culture strategy” to use Escherichia coli to efficiently produce the jasmine flavor molecule benzyl acetate. This strategy not only reduced the by-product concentration by 3 times, but also saved production time and production costs.
The authors of the report are Nika Sokolova and Kristina Haslinger from the School of Pharmaceutical Research at the University of Groningen in the Netherlands. Their main research interests are chemistry and pharmaceutical biology.

In this study, because E. coli itself does not naturally produce this compound, the researchers designed two novel biosynthetic pathways by using enzymes from different organisms with known properties. These pathways include upstream and downstream modules capable of converting E. coli’s natural aromatic metabolites into benzyl acetate.
Before testing the new downstream module, the researchers first evaluated the toxicity of benzyl acetate to E. coli and implemented a two-stage extraction culture method to ensure that E. coli remained active within the expected yield range.
Through a series of experiments, the researchers demonstrated that under specific conditions, E. coli can produce benzyl acetate. They found that when E. coli expresses only the downstream module, benzyl acetate can be produced by adding precursors. At the same time, they tried co-culturing two strains of E. coli, one responsible for the upstream module and the other for the downstream module, so that benzyl acetate could be produced from glucose.
In the process, the researchers found that the basic design of the module, the selection of enzymes in the downstream modules, and the genotype of the E. coli chassis will all affect the production of benzyl acetate. However, the inoculum ratio of the two strains in co-culture had the most significant impact on yield.
Computer simulations showed that if a single E. coli strain could express both modules simultaneously, it might outperform a co-culture system. But when the researchers tried to generate a strain of E. coli that used benzoic acid as an intermediate, they were unsuccessful. For designs with benzyl alcohol as an intermediate, they required additional genetic modification of the strain to achieve the desired yields.
Therefore, the researchers chose a co-culture system of two strains (Bn1 and Bn-BnAc3) with the best expression of the benzoate-dependent pathway module for fermentation optimization to further increase the production of benzyl acetate.
However, a large amount of ethyl cinnamate was also produced as a by-product due to the promiscuous activity of acetyltransferases during the co-culture process. To solve this problem, researchers proposed a delayed co-culture strategy. This strategy was performed in two steps, first inoculating the Bn1 strain alone to produce the target amount of benzoic acid; 48 hours later, the Bn-BnAc3 strain was added to the bioreactor to convert the newly formed benzoic acid into benzyl acetate.
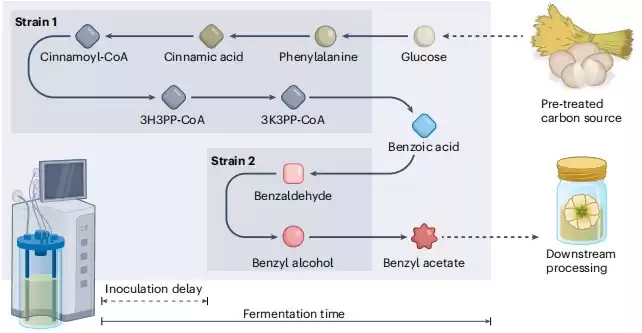
Experiments have shown that compared with the classic continuous culture strategy, the delayed co-culture strategy reduces the concentration of by-products by 3 times, and the latter uses the cell-free culture supernatant of strain 1 as the culture input of strain 2, which can save time and cost of removing cells, and may benefit from synergistic effects within the microbial flora. After further optimizing the inoculation delay, inoculum volume and density, after two fermentations of 108 hours, the concentration of benzyl acetate reached 2238±171.9 mg/L, while the production of by-product ethyl cinnamate was 797.8±220.9 mg/L. L.
Finally, the researchers assessed the economic feasibility of the process. The analysis shows that while the current costs of infrastructure and heating and cooling will be quite high, its economic viability is expected to increase as product throughput and recycling rates increase. In addition, the advantages of biosynthesis will also be more widely recognized when other factors are taken into account, such as the significant reduction in greenhouse gas emissions of biosynthesis when compared with chemical synthesis and the sustainability of its production.
To sum up, the success of this research not only provides a new and sustainable source of raw materials for perfume, cosmetics and other industries, but also provides new ideas and methods for the production of other fragrance substances, which is important for protecting the ecological environment. , Promoting industrial development is of considerable practical significance.
