How to cultivate NK cells?
Ⅰ.Preliminary preparation
1. Preparation of CulLymNK-CCK4 solution.
2. Complete medium preparation for NK activation.
3. NK amplification completes medium preparation.
Ⅱ.Flask Antibody Coating
1. Preparation of coating solution: Mix CulLymNK-CCK1 and CulLymNK-CCK5 thoroughly according to a certain ratio.
2. Add the amount indicated in the table below into the corresponding culture carrier container, and shake gently to make the coating solution evenly cover the bottom of the bottle. After that, place it horizontally in a refrigerator at 4°C and keep it away from light. It can be used after 12-18 hours of coating, and can be stored for a week after coating. For emergency use, it can be kept at 37°C, and it can be used after 2 hours of coating. (Note: After the culture bottle is coated, it is recommended to use it as soon as possible to ensure the effect of use)
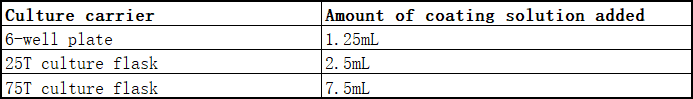
3. Aspirate the coating solution before use, and wash the coating bottle once with 7mL Buffer.
(Note: When adding Buffer to wash the coated bottle, the culture bottle needs to be erected, and the liquid is slowly added from the bottom of the bottle. Do not blow the surface of the packaged quilt, so as not to cause loss of antibodies bound to the bottle. Put the culture medium gently Pour down, let stand for 2min, then gently shake the coated bottle to elute the unbound antibody, and suck out the washing solution.)
Ⅲ.Operating procedures
DAY0
1. Sample plasma separation and inactivation
1.1. Turn on the water bath and set the temperature to 56°C.
1.2. 50mL~100mL of peripheral blood from the donor (of which 10mL is used for blood routine and infectious disease detection), and the collected blood sample is transferred to a 50mL centrifuge tube.
1.3. Centrifuge at 700g for 10 minutes (fast rise and slow fall).
1.4. Use a pipette to draw the upper light yellow plasma into a new 50mL centrifuge tube and label it.
1.5. Seal the marked centrifuge tube with parafilm, put it in a 56°C water bath, and heat inactivate it for 30 minutes.
2. PBMC isolation
2.1. Use a pipette to mix the blood cells in the lower layer with normal saline at a ratio of 1:1, and slowly add the blood cell suspension into the centrifuge tube containing Ficoll along the wall of the centrifuge tube at a ratio of 2:1 (blood cell suspension: Ficoll). (Note: operate gently, do not break the Ficoll interface)
2.2. Adjust the lifting speed of the centrifuge to the minimum, and centrifuge at 700g for 20min.
2.3. After centrifugation, add normal saline to continue centrifugation and wash twice to remove impurities such as platelets.
3. NK specific activation
3.1. Prepare complete NK cell activation medium in advance and preheat to 37°C for use.
3.2. PBMC pre-treatment (only for single-collected PBMC samples, other samples do not need to perform this step)
3.2.1. Adhesion treatment: Generally, 1×108 cells of PBMC were added with an appropriate amount of serum-free medium for NK cell activation, resuspended and left to adhere to the wall for 2 hours.
3.2.2. After the wall-attachment treatment, collect the unattached PBMC into the centrifuge tube, wash the culture bottle twice with normal saline, collect all the cell suspensions into the centrifuge tube, centrifuge at 400g for 10min, and discard supernatant.
3.3. Resuspend PBMC with NK cell activation complete medium, take a sample and count.
3.4. Transfer the cell suspension to the antibody-coated T75 culture flask, and add NK cell activation complete medium, inactivated autologous serum, and CulLymNK-CCK2 according to the cell count results.
Day 3
1. Put the complete NK activation medium into a 37°C incubator for preheating.
2. Take out the cultured NK cells, and add a certain volume of NK activation complete medium according to the growth state of the cells.
Day4
1. Put the complete NK activation medium into a 37°C incubator for preheating.
2. For NK microscopic observation and sampling and counting, an appropriate amount of NK activation complete medium can be added according to the cell density.
Day5
1. Collect the cultured NK adherent cells, centrifuge, take a sample and count, transfer the bottle to change the liquid, and continue to culture.
Day7~D14/D21
1. On day 7, put the complete NK amplification medium into a 37°C incubator for preheating.
2. Take out the cultured NK cells, observe under the microscope and count the samples, and add complete NK expansion medium to continue the culture.
3. After that, take out the cultured NK cells every other day, microscopically observe and count samples, and add NK expansion complete medium at a density of 0.8~1×106/mL to continue the culture.
4. Cultivate until D14, the total amount of cells can generally reach more than 2×109, and the longest culture time is 21 days. When the cell amount reaches the required amount, the cells are harvested by centrifugation. After passing the test, they can be used as required.
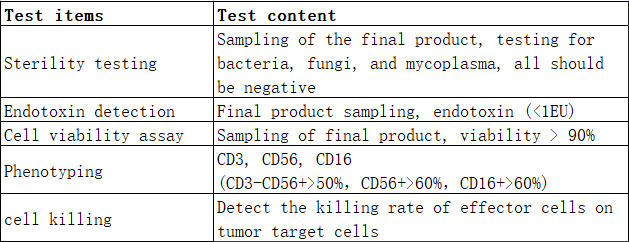
Experimental results
Ⅰ.NK cell growth curve
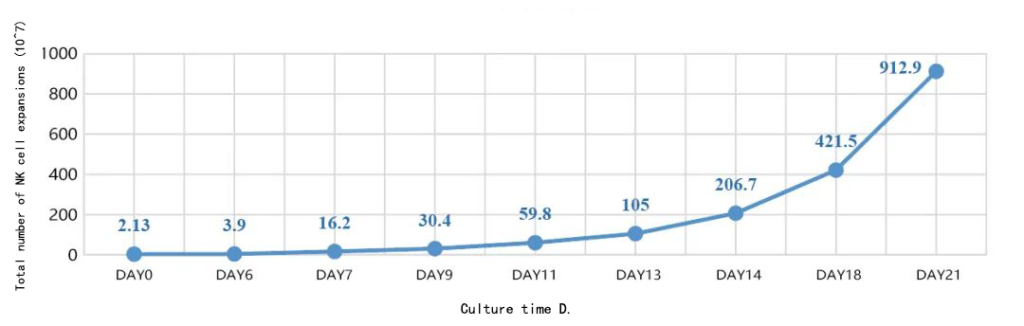
Ⅱ.NK cell flow detection
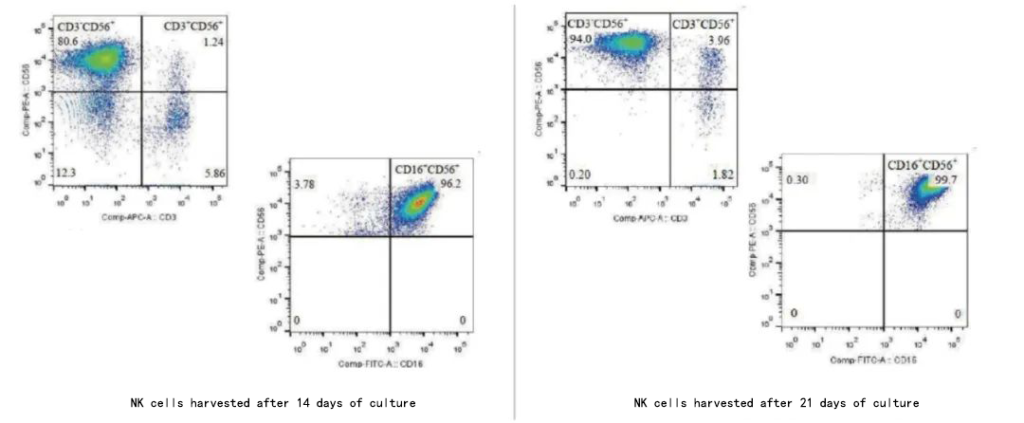
Reference Quality Inspection