The rapid development of synthetic biology technology has brought many challenges to fermentation engineering research. We need to reorganize and understand the core technologies in the optimization and amplification of fermentation processes. Optimization of the fermentation process and amplification technology are complementary to each other. If the fermentation process is developed and optimized without considering the impact of reactor scale on fermentation, it is likely that the optimized process will be difficult to apply due to the amplification effect during the amplification process. As early as the 1980s, it was proposed to reduce and reproduce the constraints of fermentation amplification based on industrial-scale reactors, and to achieve process optimization on this basis, thereby avoiding amplification failures caused by inconsistencies between the process development process and the actual production environment.
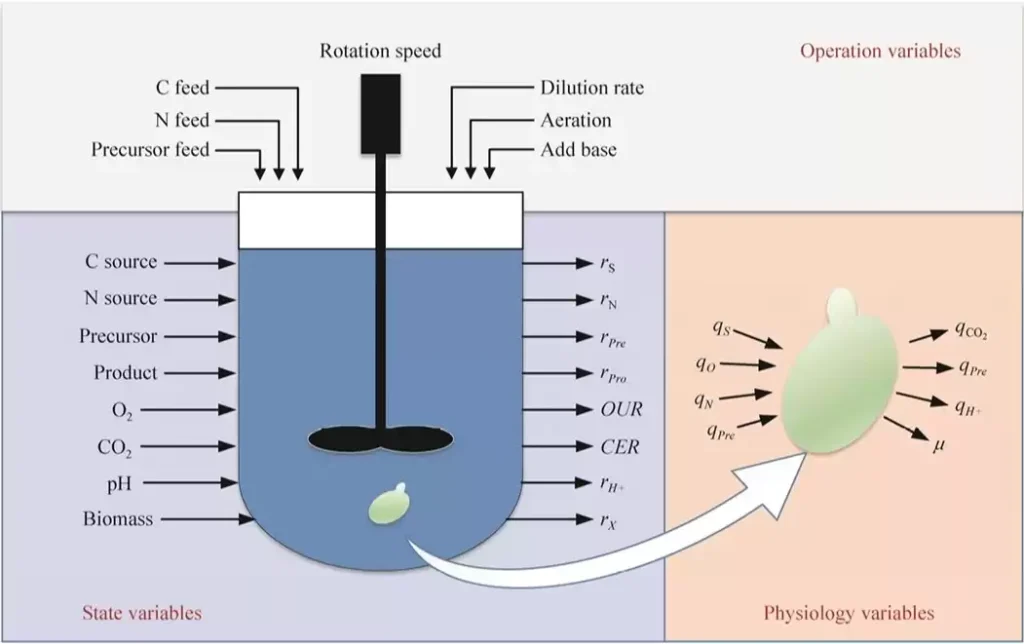
The core of fermentation process optimization is to determine the relationship between operating conditions and bacterial metabolic characteristics, and use this relationship to regulate the operating conditions so that the physiological metabolic characteristics of the bacterial cells move in a direction conducive to product synthesis, thereby achieving the optimization goal. As shown in the figure, the fermentation process carried out in the reactor involves human-operated operating parameters (operation variables), reactor-level state parameters (state variables), and cell-level physiological property parameters (physiological variables). However, due to the complex physical and chemical processes in the fermentation process, it is difficult to accurately obtain the quantitative relationship between these parameters. Currently, this goal can be achieved through different methods and strategies in order to regulate the physiological and metabolic characteristics of microorganisms to achieve high yield of target products, including multi-parameter correlation analysis methods of the fermentation process, and methods based on quantitative description of models.

The theory and method of multi-parameter correlation analysis in the fermentation process were proposed by the team of Professor Zhang Siliang of East China University of Science and Technology. They pointed out that there is a coupling correlation between various parameters in the fermentation process, which can be attributed to physical and chemical correlation and biological correlation. The physical and chemical correlation of the fermentation process mainly involves parameter correlation caused by changes in pure physical and chemical properties of substances. These physical and chemical processes include correlations caused by material or energy transfer, mixing, ion dissociation equilibrium, etc. For example, increasing the stirring speed will increase the oxygen transfer rate, and the corresponding dissolved oxygen (DO) level will increase; an increase in tank pressure will increase the concentration of dissolved CO2, causing carbonic acid to dissociate in the direction of increasing H+ concentration, thereby causing the pH value to decrease. The biological correlation of the fermentation process refers to the coupling correlation between parameters caused by cellular life activities. For example, cells grow rapidly in the early stage of fermentation, and the exponential increase in oxygen consumption rate leads to a rapid decrease in DO levels. In the later stage, the viscosity of the fermentation liquid decreases, resulting in an increase in oxygen transfer coefficient and an increase in DO levels. The most typical one is the basic correlation characteristic between cellular oxygen consumption rate (oxygen uptake rate, OUR) and dissolved oxygen: Under normal circumstances, the changes in OUR and DO curves have an inverse relationship, but when DO decreases below the critical value, oxygen supply is increased. OUR will show the same trend as DO. The critical oxygen level of microbial metabolism can be determined to a certain extent through the relationship between OUR and DO curves, so as to avoid conditions below critical oxygen during process development and optimization. In the fermentation process optimization, these physical, chemical and biochemical processes are intertwined and intricate. Methods based on correlation analysis of fermentation process parameters play an important role in the optimization process of multiple industrial fermentation products.

Industrial fermentation optimization processes usually use limiting nutrient supply rates to regulate the balance between cell growth and product synthesis, which is especially important for the synthesis of secondary metabolites. Under conditions of substrate limitation (mostly carbon source limitation) in cells, there is an interdependence between the inflow and outflow ratios in the intracellular metabolic network, that is, flux coupling. Under this condition, the absorption substrate is mainly used to synthesize bacterial cells, synthesize products, and maintain bacterial cells. In order to determine the quantitative relationship between the inflow and outflow of these flows, a process model needs to be established to describe it.
The key technical point for successful amplification of the fermentation process is consistent with the optimization of the fermentation process, that is, how to determine the relationship between the flow field environment in the reactor and the metabolic characteristics of the cells. The traditional fermentation process amplification based on a single amplification criterion encounters various problems in practical applications, which are mainly reflected in the amplification effect caused by oxygen supply capacity and uneven mixing. Theoretical analysis proves that the consistency of three or more parameters cannot be guaranteed at the same time during the amplification process. Therefore, more research focuses on the integration between reactor flow field and cell physiological metabolic characteristics. The most representative one is the fed-batch culture process of E. coli in a 22 m3 industrial-scale reactor studied by Enfors et al., which mainly analyzed the metabolic response of E. coli to a non-uniform substrate concentration field.

By using computational fluid dynamics (CFD) simulation, the description and statistics of complex flow field characteristics in large bioreactors can be achieved, which becomes the link between macroscopic bioreactor amplification design and local microscopic cell growth environment changes and cell physiological metabolism changes. important tool. In industrial practice, since fluctuations in the concentration of various nutrients will cause changes in the production efficiency of industrial processes, tracking the movement paths of cells with fluids in large bioreactors is a very valuable tool for evaluating the fermentation performance of production tanks. The “lifeline” or “movement trajectory” of cells in industrial-scale bioreactors can be obtained through Euler-Lagrange CFD simulation. This method can be further combined with metabolic models to not only provide information on the movement trajectories of cells caused by environmental fluctuations. Statistical analysis can also provide a global assessment of heterogeneous metabolic reactions within industrial bioreactors.
