Introductory
KLa (volumetric dissolved oxygen coefficient), an important indicator to characterize the oxygen transfer efficiency of biological reaction processes. For many products, there is a close correspondence between the oxygen transfer coefficient and the fermentation results, and therefore, using Kla as an amplification criterion has become a widely accepted view in the biochemical community.
It is not only the performance of the equipment that affects the KLa metrics, but also the fermentation process itself, as it has been shown that fermentation temperature, fermentation broth viscosity, air flow rate, tank pressure, and other metrics are also indicators that affect KLa. Thus KLa is clearly not a static value, but a time state variable.
Among the fermentation parameters, dissecting the concepts of dissolved oxygen, tail gas oxygen content, KLa, respiration intensity of cells, OUR, OTR and their interrelationships may be very meaningful to many newcomers to fermentation.
This paper introduces the KLa measurement method as the main line, interspersed with the discussion of the significance of the above parameters and interrelationships, the point of view may not be correct, please have different views of engineers or researchers to point out.
KLa measurement principle
Currently, the commonly used methods for KLA determination are direct method, dynamic method with sodium sulfite oxidation. This paper focuses on the use of sodium sulfite to simulate cellular oxygen depletion, combined with the homeostasis of continuous culture, which is simple and fast for the determination of KLA values.

In the presence of catalyst cobalt or copper ion ions, sodium sulfite can be rapidly oxidized to sodium sulfate. Using this feature, under the condition of continuous aeration and stirring of the reactor, sodium sulfite can be continuously injected into the reactor at a certain rate, at which time the rate of oxygen consumption can be calculated by the rate of injection of sodium sulfite
With the continuous addition of oxygen consuming agent, the dissolved oxygen in the reactor is consumed and the dissolved oxygen value decreases, thus forming the oxygen transfer driving force of the liquid phase in the reactor. In the steady state of continuous culture, the dissolved oxygen concentration in the reaction liquid is constant, i.e., dc/dt is zero, so the oxygen supply and oxygen consumption are equal at this time. Thus, the formula for KLa is obtained after the transformation of Eq:
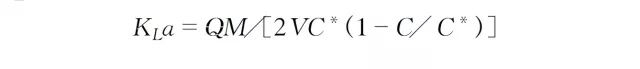
Experimental condition
Testing instruments and equipment: 10L reactor, dissolved oxygen electrode, peristaltic pump, overflow device, temperature control system and recording system are assembled into an experimental system. Different oxygen supply conditions were provided by adjusting the stirring speed and aeration volume. A 0.05 mol/l sodium sulfite solution freshly prepared with distilled water was added as the oxygen consuming agent, respectively, and the change of dissolved oxygen concentration in the reaction solution was monitored by the dissolved oxygen electrode to maintain a constant volume of reaction solution with an overflow tube. Since temperature affects the results, consistent temperature was ensured throughout the process.

Operating Methods
In the experiment, a certain amount of 7L distilled water was added to the reactor, then 5ml,1mol/l CuSO4 solution was added to the reactor, and continuous aeration was carried out for 15min, so that the dissolved oxygen in the reaction solution reached the saturation state. The output of dissolved oxygen electrode was calibrated to 100% and recorded automatically. The newly prepared sodium sulfite solution was added into the reactor by peristaltic pump, the aeration volume was adjusted by flowmeter valve, and the stirring speed was adjusted between 100~800rpm. According to the difference of oxygen supply capacity, change the rotational speed of peristaltic pump accordingly, so that under each operating condition, a steady state can be achieved. After the dissolved oxygen concentration was constant, the C/C* value was recorded.
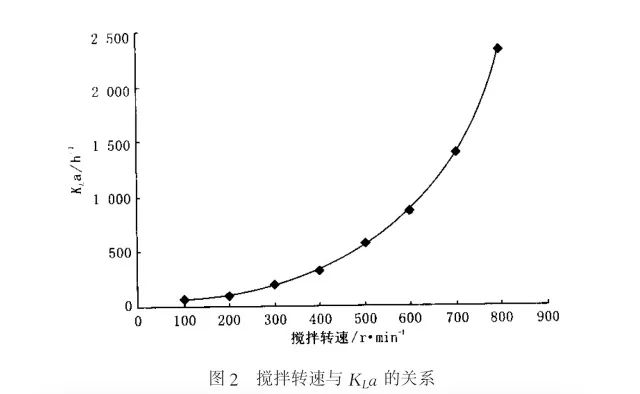
A dynamic approach to calculating KLa
OTR is the capacity or state of the facility to supply oxygen in fermentation and OUR is the level or state of cellular oxygen uptake. Thus OTR is equivalent to the product of KLa and C*-C. So when DO rises, it means that OTR is greater than OUR, and the same is true for the reaction.
When the oxygen content of the exhaust gas can be detected, the OUR can be calculated by conservation of mass through the air flow, air oxygen content and exhaust gas oxygen content. dc/dt can be calculated from the change in dissolved oxygen. For example, if the DO decreases from 70% to 60% in 1 hour, the change in oxygen concentration equivalent to 10% DO can be calculated. c*-C can also be calculated from the change in oxygen concentration equivalent to 35% DO (the mean value of 60% and 70% is 65%, and the difference from 100% is 35%). In this way KLa can be calculated dynamically, then OTR, can be obtained from KLa and C*-C.
Discussion and tips
1.DO is generally in the fermentation process, expressed as a percentage, and if it is not calibrated using absolute concentrations, then it is not possible to perform dynamic calculations. It is believed that some software carries out default settings, otherwise it is not possible to get online KLa values.
2.Generally enterprises set a minimum DO value as a reference for critical oxygen concentration, or set a reference value for normal DO and adjust it when it deviates.
3.I believe that through the description of the dynamic KLa measurement in this paper, you have a clear idea of the relationship between OUR,KLa, C*, and the significance of these parameters. So even if you are not able to measure other parameters and only have a DO, you should be able to take into account the changes in other relevant parameters. Only then will it be possible to develop a rational regulation program and a valuable interpretation of the program.
4.There are already articles that analyze the above algorithms in more depth, such as the effect of temperature on OTR, and the effect of water content in the air on OTR. For a deeper understanding of these parameters, please find these articles to study.
Reflections:
1, there is a more worthy of study is: is when the DO drop, take which means more reasonable? For example, you can change the air flow, mixing speed, tank pressure, replenishment of water, with release, etc. I believe that any literature is not possible to have an operational description of this issue, the need for engineers to develop their own plans in conjunction with the actual situation.
2. how to explain the effect of different seasons on the variation of OTR?
