Preface
293T cells are widely used for transient transfection to overexpress various recombinant proteins or viral packaging, and their advantages include high transfection efficiency, low-cost amplification, and human glycosylation patterns [1]. With the rapid development of gene therapy and cell therapy, 293T cells are increasingly used in adeno-associated virus (adeno-associated virus, AAV) vectors, recombinant adeno-associated virus (rAAV) vectors and lentivirus production[2]. In order to achieve larger-scale GMP production requirements to meet the research and development and clinical experiments related to gene therapy and cell therapy, more and more researchers are turning their attention to bioreactor technology.
There are three major challenges in the process from incubator to bioreactor. First of all, 293T is originally an adherent cell and needs to attach to the surface of the carrier to grow. Currently, the most commonly used adherent carrier is the Cytodex1 spherical carrier. For example, the cell density of Vero cells can reach more than 1.0×107 cells/mL in this system. However, the adhesion firmness of 293T cells is weaker than that of adherent cells such as Vero cells. During the stirring process of microcarriers, cells will fall off and die, so there are still limitations in production applications. Secondly, adherent 293T cells need to add serum and other animal-derived components during the growth process, which affects later quality control. Finally, adherent cells are limited by the efficiency of cell digestion during step-up scaling. Faced with these challenges, some researchers have domesticated 293T cells into a serum-free suspension cell line to solve the above problems, but the cell density can only be at the level of 106 cells/mL [3].
As a favorable tool for large-scale animal cell culture, bioreactors have been applied to the 293T suspension culture process. However, in the usual batch mode, the depletion of nutrients and the accumulation of metabolic by-products often limit cell growth and metabolism. The fed-batch culture process has become an animal The mainstream model for large-scale cell culture. Therefore, we used 293T suspension cells for Fed-batch culture on the CloudReady™ 500mL bioreactor system to provide a reference for high-density expression of 293T suspension cells (Figure 1) and provide new feasibility for the viral vector production process.

Figure 1 500mL Bioreactor System
Operation process
In this experiment, a cell density of 5×105 cells/mL was used to inoculate, and air and oxygen were cascaded into DO, CO2 and 0.5 mol/L NaOH solution cascade pH. See Table 1 and Table 2 for other parameter control and feeding control. In addition, on the eighth day, an additional 10 mL of feed A for transient expression of 293 and 1 mL of feed B for transient expression of 293 were added based on the actual conditions of the cells.
Table 1 Process control parameter settings
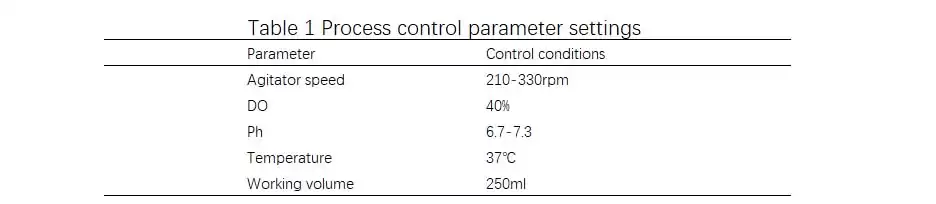
Table 2 Feed control conditions

Result
This experiment carried out a 217h culture period. After controlling the above conditions, the final cell density reached 2.28×107 cells/mL, and the viability rate was maintained above 93% (Figure 2). During the experiment, no 0.5 mol/L NaOH solution was introduced, and the lowest pH value was maintained above 6.7 (Figure 3). According to the experience of CARE (Debier Biotechnology and Engineering Research Center) in the early stage of culturing 293T cells, controlling the pH at 6.9-7.3 will cause a large amount of lactic acid to accumulate, which will continue to increase to 4.41g/L during the culture period [4]
In this experiment, the pH setting value was lowered, and the maximum lactic acid concentration did not exceed 2g/L, and finally stabilized at around 1.5g/L (Figure 4). Such results provide a certain reference for the next step of process optimization using micro-parallel bioreactors.
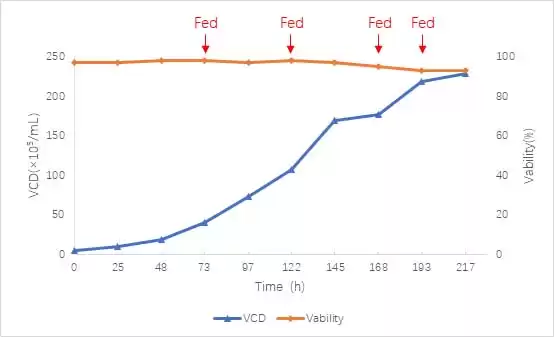
Figure 2 Proliferation curve of 239T cell culture process, “Fed” refers to the two media supplemented with feed A and B for transient expression of 293.
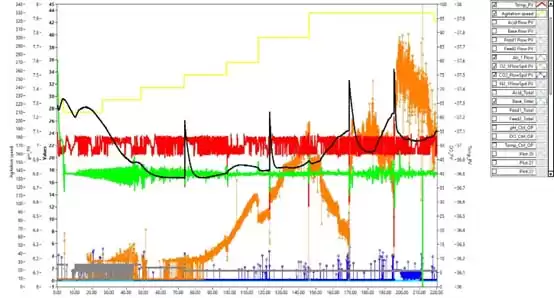
Figure 3 293T cell culture cycle parameter control trend chart: red – temperature; black – pH; green – DO; yellow – rotation speed (rpm); blue – CO2 flow rate (mL/min); gray – air flow rate (mL/min) ; Orange – Oxygen flow rate (mL/min).
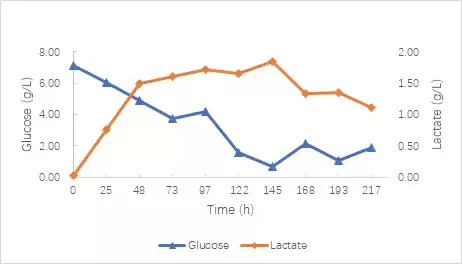
Figure 4 Glucose consumption curve and lactate metabolism curve during 239T cell culture.
