This article focuses on a comprehensive summary of the development of vaccine process technology over the past decade, including the emergence of new vaccine platforms, approvals of various types of vaccines, advances in production technology, and improvements in analytical characterisation capabilities.

I. Progress in vaccine development and approval
1. New platform vaccines approved
Vaccines containing DNA and RNA (viral vectors and mRNA-LNP vaccines) are developing rapidly, with ten new viral vector vaccines (e.g., against Japanese encephalitis, dengue, Ebola, SARS – CoV-2, etc.) and two mRNA-LNP vaccines approved since 2012.
Some viral vector vaccines use special strategies, such as the heterologous strategy (GamEvac – Combi), where different vectors (e.g. VSV/Ad5) are used to carry the antigen.体(如VSV/Ad5)
2. New developments in traditional vaccines
In 2012 Flucelvax became the first U.S.-introduced seasonal influenza vaccine produced in cell culture technology using suspension MDCK cell culture, which had been approved in Europe in 2007.
In 2017 the FDA approved a variety of vaccines such as Shingrix, a shingles vaccine (recombinant glycoprotein antigen, produced in CHO cells with specific adjuvants), and Hepatitis B vaccine Heplisav – B (with novel adjuvants and recombinant protein subunit antigens).
FDA approval of the smallpox and monkeypox vaccine JYNNEOS (a live vaccine produced from the MVA – BN strain, grown in specific cells and processed through a multi-step process) in 2019, and its administration in 2022 due to an emergency use authorisation extension.
WHO approves multiple polio vaccine-related advances in 2020, such as nOPV2 (a modified strain prepared using Vero cells with genetic modifications to improve stability) and sIPV (microcarrier-based Vero cell production with several improvements to reduce risk).
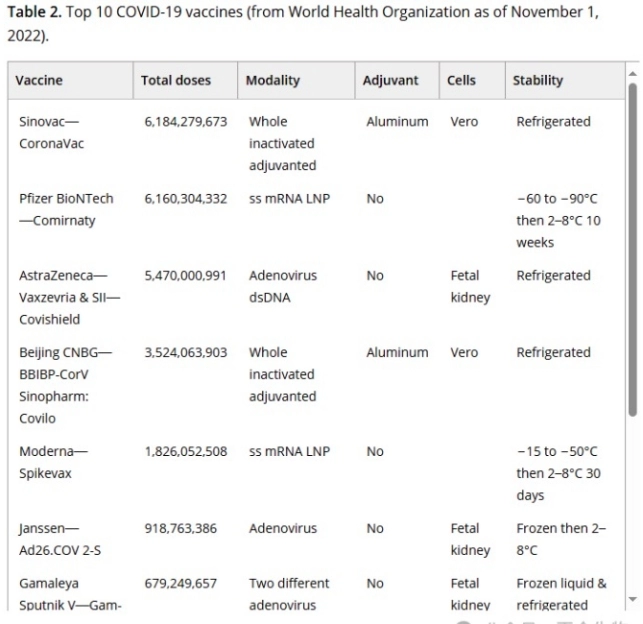
3. COVID-19 vaccine results
Following the COVID – 19 outbreak in early 2020, numerous vaccine candidates entered clinical trials, with the first EUAs awarded in December of the same year, and by March 2023 over 13 billion doses had been administered.
The main vaccine modalities include whole virus inactivation (e.g., Coxing CoronaVac, Sinopharm BBIBP – CorV, etc.), mRNA – LNP (e.g., Pfizer BioNTech Comirnaty, Moderna Spikevax) and adenoviral vectors (e.g., AstraZeneca Vaxzevria, Concinol Ad5 – nCoV, etc.), and different vaccines have their own features on the number of doses, adjuvants, cells, stability, etc. have their own characteristics.
II. Technological advances in vaccine production
1. Cell culture
Adherent Cell Lines
The selection of cell lines for vaccine production has changed over time, from limited to increased use of continuous cell lines based on safety concerns.
For example the rVSVΔG – ZEBOV – GP Ebola vaccine is produced in Vero cells with a specific modification of the virus strain.
Innovative developments in disposable bioreactors, such as the SUB with its large surface area per unit volume, and fixed-bed reactors combine enhanced continuous and automated disposable bioprocessing methods. Polio vaccines embody a complete process linkage from upstream cell culture, downstream processes to inactivation.
suspension culture
Various vaccine applications suspension cultures, such as baculovirus-expressing insect cell culture (Sf9) are used for large-scale production of influenza vaccine HA proteins.
Cells such as Vero or PER.C6 cells can be domesticated and then cultured in suspension for virus production, and improved CHO cell culture techniques (such as the SARS – CoV – 2 vaccine candidate described by Watterson et al.) can be applied to vaccine production, including VZV-truncated glycoprotein E for the production of Shingrix vaccines.
COVID – During the 19th pandemic, the University of Oxford successfully scaled up vaccine development based on a replication-defective adenovirus platform, using suspension cell cultures expressing adenoviruses to deliver spiny proteins to supply large quantities of vaccine.
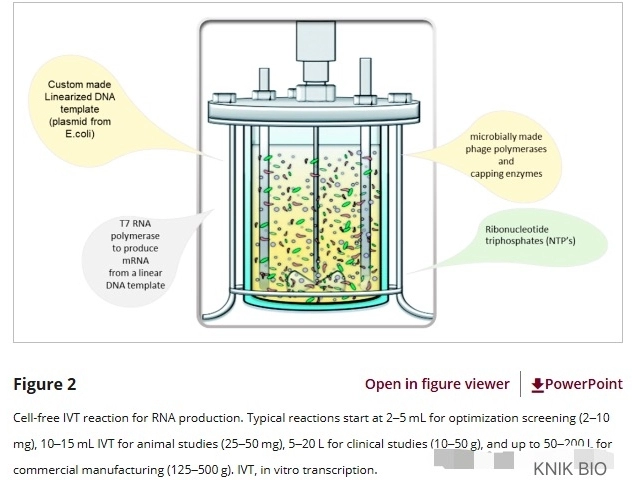
2. Microbial production
Traditional applications and new developments
Microbial fermentation platforms are used to produce a wide range of vaccines, e.g. Saccharomyces cerevisiae expresses the L1 protein of HPV assembled into a VLP to produce the Gardasil vaccine, Hepatitis B Virus Surface Antigen can be expressed by either Saccharomyces cerevisiae or Picrospiracid yeast, and Pneumococcal Conjugate Vaccines are conjugated from the expression of components by specific strains of the organism.
New cultures are advancing, such as the Dyadic platform based on Thermothelomyces heterothallica fungal cultures, which allows rapid development of stable strains, high yields, easy scalability and cost-effectiveness, and whose expression of the COVID – 19 vaccine candidate is under clinical evaluation. MIT’s automated platform based on Picros yeast produces a wide range of proteins, including vaccine antigens, and is currently used for rotavirus vaccine candidate production.
For protein antigen production, there are challenges in producing vaccines that expose key epitopes, E. coli fermentation is the main method for producing pDNA templates for mRNA vaccine production, there are advances in cell-free synthesis of pDNA but cost-effectiveness needs to be improved and there are relevant considerations for pDNA production in terms of strain selection and sequence characterisation, and new conjugate vaccine production methods are also emerging.
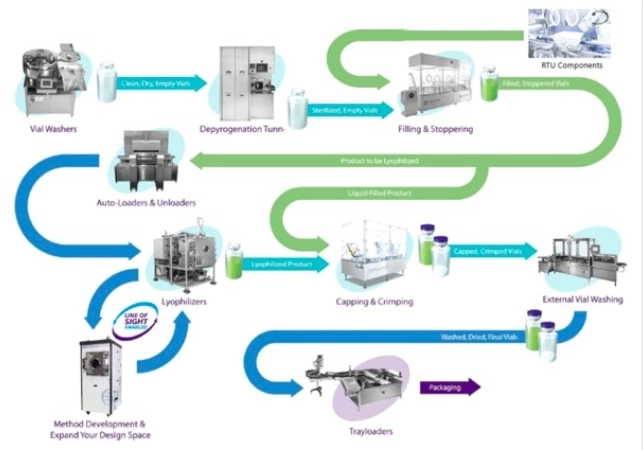
Process operations for liquid and lyophilized products. Image adapted with permission from ATS Scientific product.
III. Summary and outlook
The past decade has seen significant advances in vaccine technology, including the development of new platforms, the approval of various vaccines, innovations in production techniques and improved analytical characterisation capabilities, which have important implications for the prevention of infectious diseases and future therapeutic oncology vaccines, and the field of vaccine research and development will continue to bring together new technologies, methodologies and platforms for the betterment of human health in the future.
KNIK BIO has a wealth of experience in the manufacture of various types of reactors and pressure vessels. With a team of experts in the fields of biological reaction, fermentation process, machinery manufacturing and automation control, our scientific research and technological level are always in the leading position in China and the international first-class level, which provides you with a comfortable, assured and safe product experience.
