★L-tryptophan is an essential amino acid widely used in food, medicine and feed production industries. At present, the engineering strains used to produce L-tryptophan through biological fermentation are mostly Escherichia coli and Corynebacterium glutamicum, which are mainly obtained through rational methods such as metabolic engineering and synthetic biology. However, due to the long metabolic pathway of L-tryptophan in microbial cells and the complex and unclear regulatory mechanism, production strains still have problems such as low production efficiency and poor robustness. It is often necessary to transform and improve strains through irrational methods such as adaptive evolution. production capacity.
In order to construct a strain that efficiently produces L-tryptophan, the researchers targeted the main factors that limit the synthesis of L-tryptophan and used traditional mutation breeding and various metabolic engineering strategies to systematically transform and optimize the strain. Currently, they have achieved A series of fruitful results. These modifications are mainly concentrated in bacteria such as Escherichia coli, and the modification strategies include the following aspects.
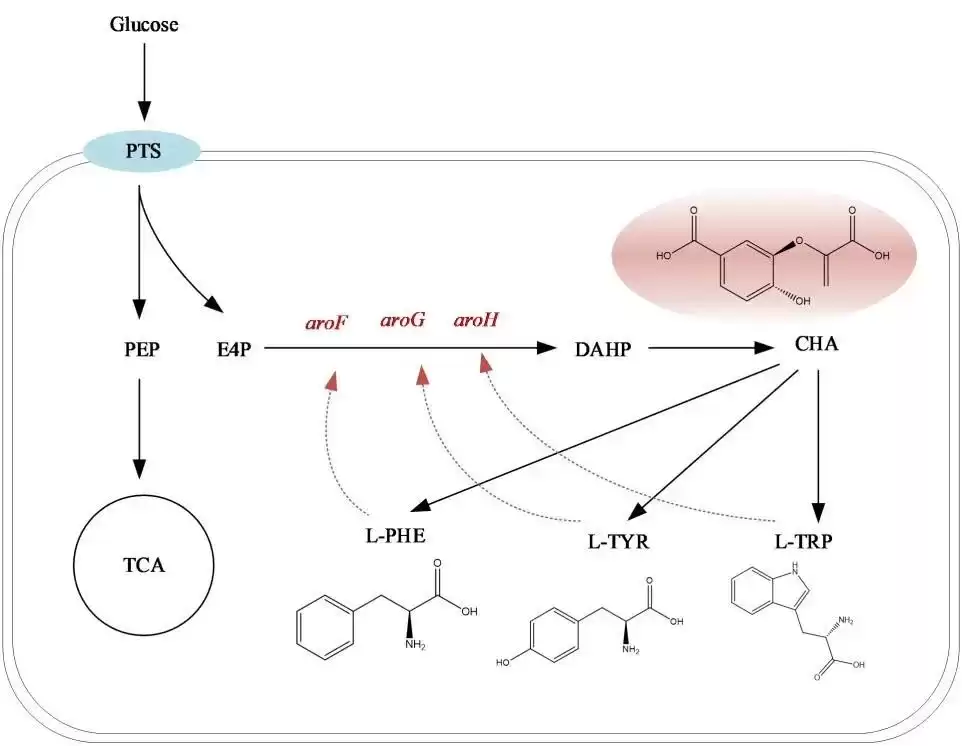
◆Enhance precursor synthesis
L-tryptophan has a multi-step and complex synthesis pathway in cells, requiring many precursor substances. However, these precursor substances are widely involved in the metabolic reactions of various substances in cells. Therefore, enhancing the synthesis of intracellular precursor substances or limiting the consumption of precursor substances in other metabolic pathways is an effective method to enhance the synthesis capacity of L-tryptophan (Figure 2). Zhu et al. increased the production of E. coli L-tryptophan-producing bacteria by 276% by optimizing the supply of precursors and adjusting the balance of cofactors; Chen Liping conducted mutagenesis screening of E. coli L-tryptophan-producing bacteria, making L -Tryptophan production reaches 55.1 g/L; Xiong Bo optimized the metabolism of Escherichia coli L-tryptophan engineering bacteria and obtained a strain with L-tryptophan production reaching approximately 42 g/L and sugar acid within 40 hours. L-tryptophan-producing bacteria with a conversion rate of approximately 0.23 g/g.
❶Promote DAHP synthesis
Increasing the DAHP synthesis rate is the key to increasing carbon flux into the common pathway (Figure 2) and is also a feasible method to promote excessive accumulation of L-tryptophan in cells.
❷Enhance enzyme activity in common pathways
In the common pathway, substances such as SHIK, EPSP and CHA are all important intermediate metabolites (Figure 2). Although there have been cases of improving the efficiency of the common pathway by increasing the production of PEP and E4P, the limited activity of a series of enzymes in the common pathway is still an important factor hindering its synthetic efficiency.
◆Enhance L-tryptophan branch pathway
After common metabolic pathways, L-tryptophan, L-phenylalanine and L-tyrosine metabolic pathways can all be used as CHA metabolic pathways. Therefore, to increase the production of L-tryptophan, it is necessary to make as much CHA flow into the L-tryptophan metabolic pathway as possible through metabolic regulation.
❶Blocks the metabolic pathways of L-phenylalanine and L-tyrosine
Blocking the metabolic pathways of L-phenylalanine and L-tyrosine not only allows more CHA to flow to the L-tryptophan pathway, but also reduces the feedback of these two amino acids in the common pathway and the central metabolic pathway. Inhibiting, weakening the expression of pheA gene and tyrA gene or reducing the activity of these two enzymes is the main method to block the L-phenylalanine and L-tyrosine metabolic pathways.
❷Unblock L-tryptophan branch metabolic pathways
In addition to removing the above two branch metabolic pathways, research has also increased the production of L-tryptophan by enhancing the L-tryptophan metabolic pathway. The main enhancement pathways include the release of feedback inhibition and overexpression of the corresponding synthetases.

◆Transform the transfer system
When the intracellular concentration of L-tryptophan is too high, it will disturb the metabolic balance of cells and cause physiological toxicity to cells. Therefore, reducing the intracellular content of L-tryptophan is one of the key factors to increase L-tryptophan production. However, since the mechanism of L-tryptophan secretion is still unclear, modifications to the transport system mainly focus on blocking the absorption pathway. In E. coli, there are three transport genes that can regulate the absorption of L-tryptophan, namely the mtr, tnaB and aroP genes. Among them, the two transport enzymes encoded by the mtr and tnaB genes regulate the absorption of L-tryptophan. The amino acids are specific and encode two permeases with different affinities. The transporter encoded by the aroP gene is not specific for L-tryptophan and can also transport L-tyrosine and L-phenylalanine. At the same time, the absorption of L-tryptophan by Corynebacterium glutamicum is completely regulated by the aroP gene. However, current research on the efflux system of L-tryptophan still fails to clearly analyze its mechanism, and the regulation method is still unknown.
◆Adaptive evolution improves the performance of L-tryptophan-producing strains
An important factor currently limiting the production level of L-tryptophan produced by direct fermentation is that excessive intracellular accumulation of L-tryptophan will cause physiological toxicity to cells, thus improving the tolerance of strains to high concentrations of L-tryptophan. Sex is of great significance to the fermentative production of L-tryptophan.
❶Tradition adapts to evolution
Adaptive evolution is a method that combines artificial selection pressure and simulates the compilation and selection process in natural evolution to achieve directional evolution of microorganisms under laboratory conditions to select individuals with excellent traits from the evolved population.
❷Biosensor binding
Biosensors are widely used components in synthetic biology, which can adjust the expression of downstream genes by sensing the concentration of corresponding substances. By combining synthetic biology components with adaptive evolution, cells can express specific traits of different strengths based on differences in intracellular tryptophan concentrations, thus providing conditions for efficient screening.

At present, a large number of attempts have been made to transform tryptophan-producing strains of Escherichia coli and Corynebacterium glutamicum, and certain results have been achieved. However, due to the unclear tolerance mechanism of strains to high concentrations of L-tryptophan, it is still not possible to better Relieving the physiological toxicity of L-tryptophan to strains, resulting in low production intensity and poor robustness of production strains, is the main difficulty encountered in the industrial large-scale production of L-tryptophan today. Although adaptive evolution has great practicality, it suffers from problems such as long time required and large labor force. Therefore, combining biosensors with adaptive evolution methods can effectively improve evolutionary efficiency, thereby conducting in-depth research on the L-tryptophan tolerance mechanism of production strains, reducing the toxic effects of L-tryptophan, and finding ways to use L-tryptophan Methods to efficiently expel bacterial cells will promote the further development of rational metabolic transformation.
